PolyMed Pharma jobs are gaining strong attention among candidates looking for high-growth careers in the medical devices and pharmaceutical industry. This walk-in drive offers multiple opportunities across engineering, quality, production, and R&D domains for experienced professionals seeking stability and advancement.
Company overview:
PolyMed Pharma jobs provide excellent career opportunities in a globally recognized medical devices manufacturing company. Poly Medicure Limited is a leading manufacturer of medical devices and pharmaceutical products with a strong presence in international markets. The company is known for innovation, high-quality standards, and compliance with global regulatory guidelines. With advanced manufacturing facilities and continuous focus on research and development, PolyMed offers employees a professional work culture, technical learning, and long-term career growth.
Job details:
PolyMed Pharma jobs include multiple openings across technical and functional departments with varied experience requirements.
Job Title:
Documentation Officer
Engineer
Senior Engineer
Project Manager
Assistant Manager
Deputy Manager
Manager
Senior Manager
Chemist
Department:
R&D, Tool Design, Moulding, Purchase, Maintenance, PPC, Automation Electrical, Automation Mechanical, Regulatory Affairs, Quality, Production, Tool Room
Salary:
N/A
Requirements:
Skills:
Knowledge of ISO 13485 documentation and regulatory compliance
Experience in software like CREO and SolidWorks
Understanding of injection moulding and plastic technology
Hands-on experience in maintenance and utilities
Knowledge of HVAC, electrical systems and instrumentation
Experience in production planning and inventory management
Understanding of PLC, HMI and servo systems
Experience in QA, QC, QMS and validation activities
Knowledge of HPLC, chemical analysis and testing methods
Strong coordination, communication and problem-solving skills
Qualification:
BTech, BE, BPharma, MPharma, BSc, MSc, Diploma, Post Graduate Diploma
Experience:
2 to 15 Years
Vacancies:
Not yet disclosed
Interview Details:
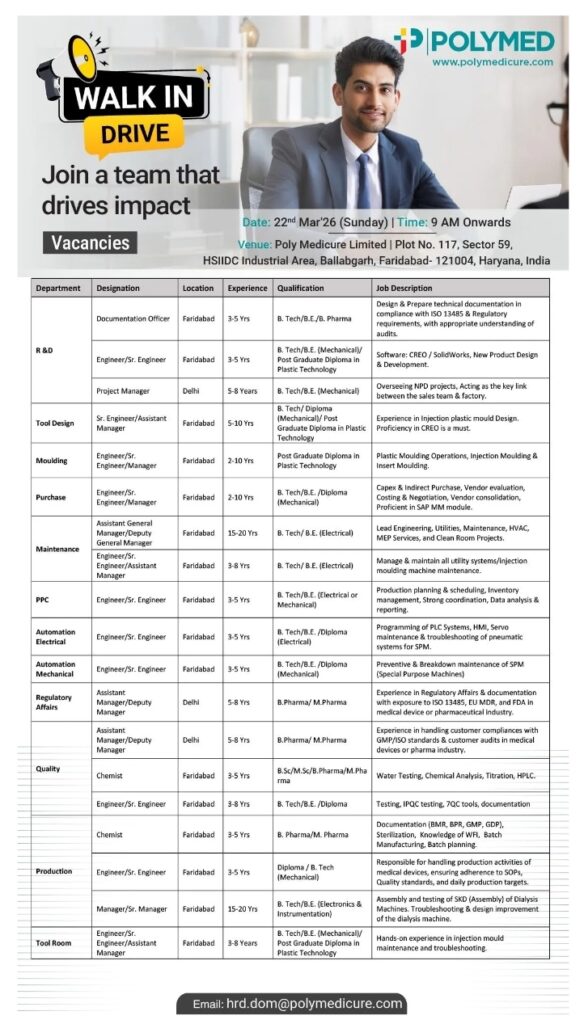
- 📁 Department: R&D
📄 Qualification: BTech, BE, BPharma
📄 Experience: 3 to 8 Years - 📁 Department: Engineering, Tool Design, Maintenance
📄 Qualification: BTech, BE, Diploma
📄 Experience: 3 to 15 Years - 📁 Department: Quality, Regulatory Affairs
📄 Qualification: BPharma, MPharma, BSc, MSc
📄 Experience: 3 to 8 Years - 📁 Department: Production, PPC, Automation
📄 Qualification: BTech, Diploma
📄 Experience: 3 to 8 Years
Type:
Walk-In Drive
Date and Time:
22nd March 2026
09:00 AM onwards
Location/Platform:
Poly Medicure Limited
Plot No. 117, Sector 59
HSIIDC Industrial Area
Ballabgarh, Faridabad, Haryana, India
Documents Required:
Updated CV/Resume
Educational Certificates
Experience Certificates
ID Proof
Recent Photograph
How to Apply:
Candidates can directly attend the walk-in drive at the mentioned venue with required documents
Candidates can also share their updated CV via email: hrd.dom@polymedicure.com
PolyMed Pharma jobs offer a strong platform for candidates who want to build a career in medical devices and pharmaceutical manufacturing. The company provides exposure to advanced technologies, global compliance standards, and continuous professional development. Candidates are advised to prepare well, revise technical concepts, carry all original documents, and maintain professional appearance during the interview. This opportunity is ideal for professionals looking for long-term growth and stability.
FAQs:
What is the experience required for PolyMed Pharma jobs?
Candidates with 2 to 15 years of experience are eligible depending on the role.
What qualifications are required for these positions?
BTech, BE, BPharma, MPharma, BSc, MSc, Diploma and PG Diploma candidates can apply.
Which departments are hiring in this walk-in drive?
Departments include R&D, Engineering, Quality, Production, Maintenance, Automation and more.
Is this walk-in open for freshers?
No, these roles are mainly for experienced candidates.
What is the walk-in interview date?
The walk-in is scheduled for 22nd March 2026.
What time does the interview start?
The interview starts from 09:00 AM onwards.
What documents are required for the interview?
Candidates must carry resume, educational certificates, experience proof, ID and photos.
What type of work is involved in production roles?
Production roles include handling manufacturing processes, assembly, and quality compliance.
Is regulatory experience required?
Yes, for regulatory affairs roles knowledge of ISO and compliance standards is required.
Can candidates apply through email?
Yes, candidates can send their CV to the provided email ID.
 Follow us on LinkedIn
Follow us on LinkedIn
